Glioblastoma
A glioblastoma is the most common high grade primary brain tumour in adults. It rarely occurs in children.
Short summary
Glioblastoma brain tumours are sometimes called glioblastoma multiforme, GBM, GBM4 or a grade 4 astrocytoma.
They’re often treated through chemotherapy, radiation therapy, and specific drugs. Surgery is also a common option depending on where the tumour is in the brain.
Below we’ll take a closer look at glioblastoma causes, symptoms, prognosis, and treatments.
On this page:
- What is a glioblastoma?
- What symptoms can a glioblastoma cause?
- How are glioblastomas treated?
- What causes glioblastomas?
- How long can I live with a glioblastoma?
Get support
It’s normal to feel shocked if you or someone you know has been diagnosed with a glioblastoma. Our Support Team can answer questions you have or provide a listening ear if you need one.
Get your free Information Pack
Our Brain Tumour Information Pack can help you better understand your diagnosis and feel confident talking to your medical team.
Get the BRIAN app
BRIAN is our trusted online app where you can track your experience, compare it with others who’ve been there and get the knowledge you need to make informed decisions.
What is a glioblastoma?
Glioblastomas are grade 4 brain tumours. Grade 4 is based on the World Health Organisation (WHO) grading system. Grade 1 and 2 tumours are usually not cancerous and can grow quite slowly. But grade 3 and 4 tumours are more advanced.
They:
- grow quickly
- are diffuse, which means they have tendrils that reach into other parts of the brain
- are likely to spread within the brain
- can come back, even if they’ve been treated
- are sometimes called malignant or cancerous.
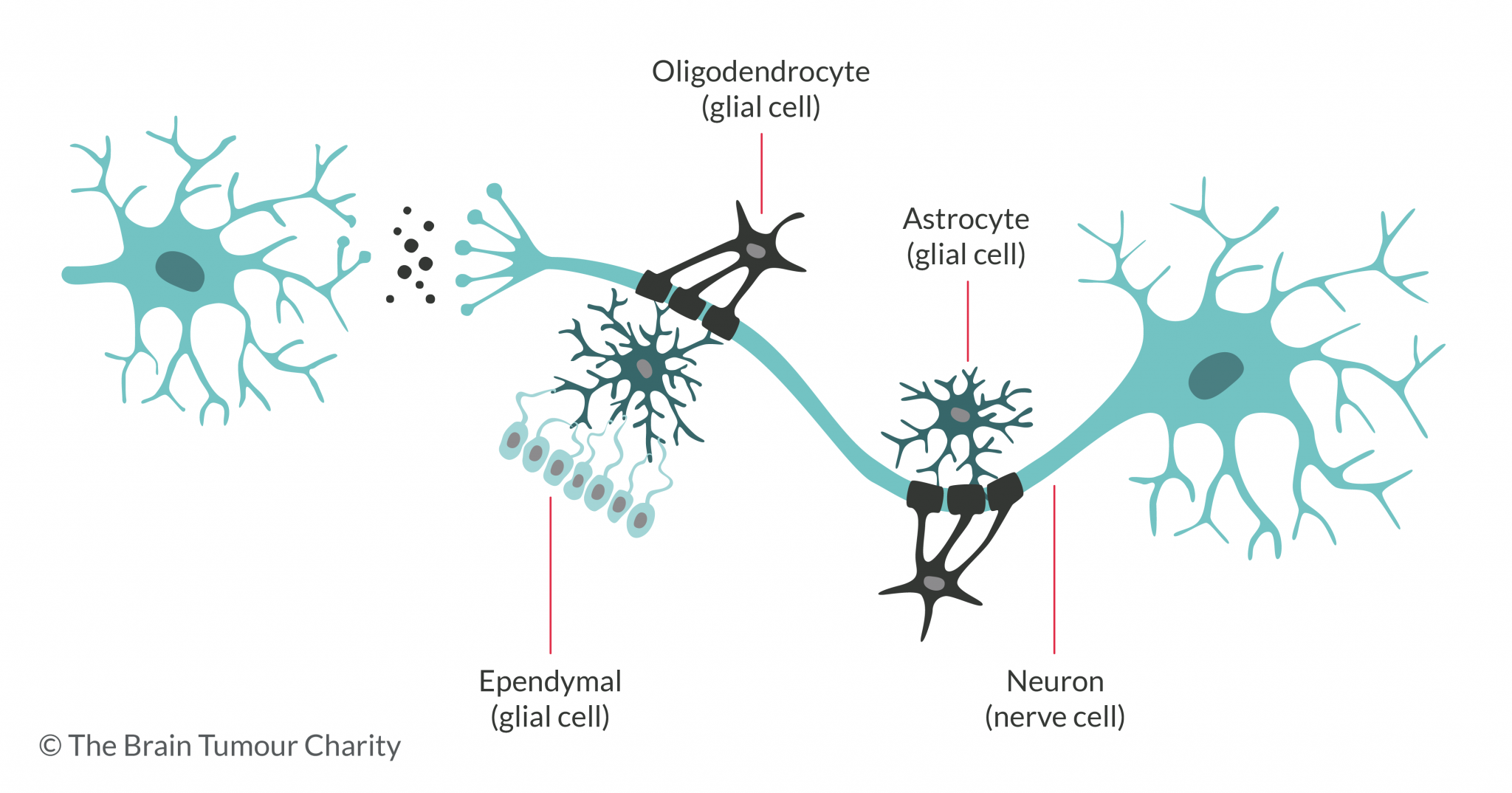
GBMs are a type of glioma, which is a brain tumour that grows from a glial cell. This is where the term, ‘glioblastoma’ comes from. The ‘multiforme’ comes from the fact that they have lots of variations, or different forms. This can unfortunately make them more difficult to treat than other tumours.
Glioblastomas form from cells called astrocytes, which are there to support nerve cells.
What are the symptoms of a glioblastoma?
Different parts of the brain control different functions, so the symptoms you experience will depend partly on where the tumour is within your brain. It will also depend on the treatment you receive.
But, some symptoms of a glioblastoma are:
- headaches, caused by pressure in the brain
- personality changes
- trouble remembering things
- trouble speaking or understanding
- tiredness
- depression
- trouble thinking
- seizures
- sight problems.
For more about the symptoms of a brain tumour, click the button below.
How are glioblastomas treated?
The treatment for glioblastoma depends on the person, where the tumour is sitting in the brain, and other specific things. But, these are common treatments:
Generally, if you’re well enough, you’ll have neurosurgery to take out as much of the tumour as possible.
Once your wound has healed, you might also have chemotherapy, radiotherapy or both.
Before surgery, you may want to ask your healthcare team about:
- 5-ALA. A surgical aid that can help surgeons remove more of a tumour
- biobanking. A method of storing a sample of your tumour for future research.
- clinical trials. Experiments into new ways of treating brain tumours.
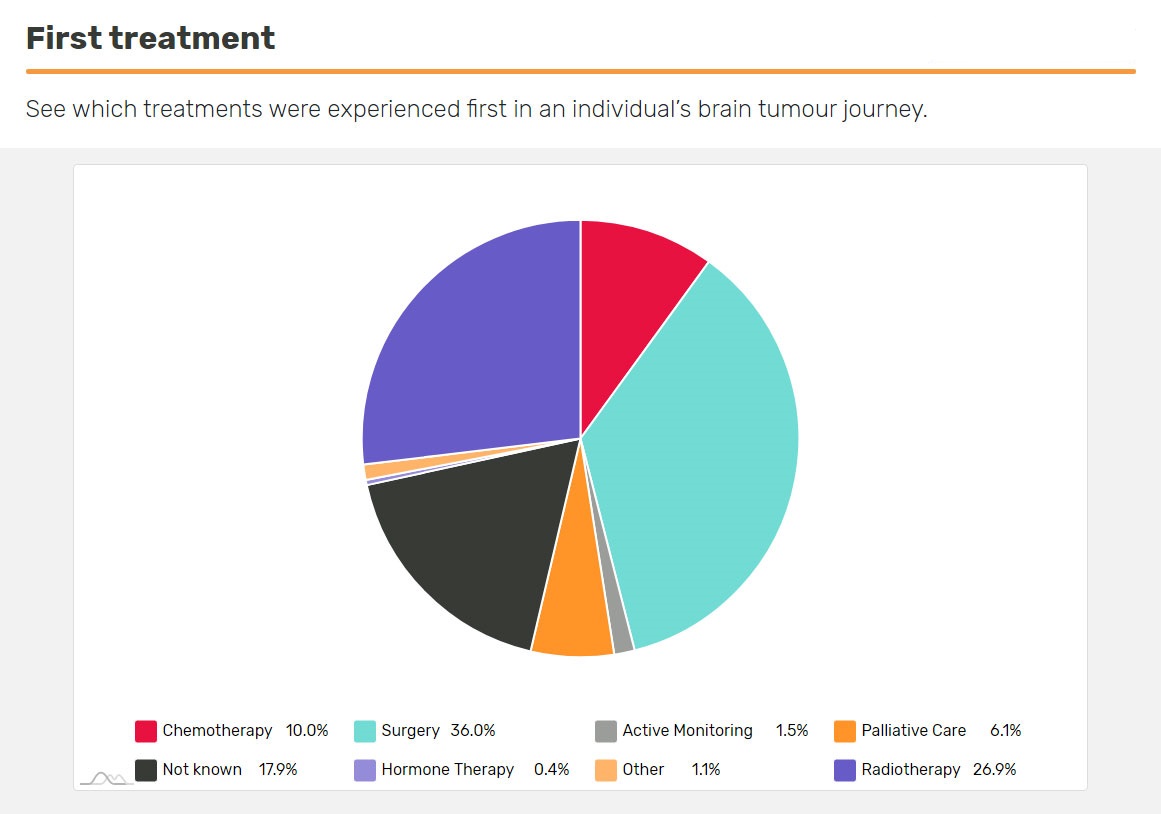
What treatment do people diagnosed with a glioblastoma usually have first?
If you’ve just been diagnosed and are about to have treatment, you may want to see what other people’s first treatment was. Use the First Treatment insight in BRIAN, which you can personalise to make it relevant to you.

Trial finds DCVax-L can prolong the lives of those living with a Glioblastoma
A major phase III clinical trial has found that novel treatment DCVax®-L can prolong the lives of people diagnosed with a new or recurrent glioblastoma
What causes glioblastomas?
As with most brain tumours, it’s not known why glioblastoma multiforme tumours start growing, although we do understand some of the risk factors involved.
It’s important to know that there is nothing you could have done, or avoided doing, that would have caused you or somebody you know to develop a brain tumour.

Glioblastoma research
Read about the research we are funding to help our understanding of how and why this tumour type forms and develop new, effective treatments.
How long can I live with a glioblastoma?
Nobody can be absolutely certain about what will happen to you following a diagnosis of a brain tumour.
Your healthcare team may give you a prognosis, which is an estimate based on your tumour type and current situation. But, they won’t be able to know things specific to you, like how well you might respond to treatment.
Click the button below to learn more about glioblastoma prognosis.
Support and Information Services
Research & Clinical Trials Information
You can also join our active online community.
In this section

Get support
If you need someone to talk to or advice on where to get help, our Support and Information team is available by phone, email or live-chat.
Recommended reading
Share your experiences and help create change
By taking part in our Improving Brain Tumour Care surveys and sharing your experiences, you can help us improve treatment and care for everyone affected by a brain tumour.